Part 1
Coursework Aim
In this part of the coursework, you will determine the mechanism and develop a rate law for a catalytic reaction from experimental data.
Question 1
Find the rate law for the following catalytic reaction based on the data provided.
𝐴𝐴 → 𝐵𝐵
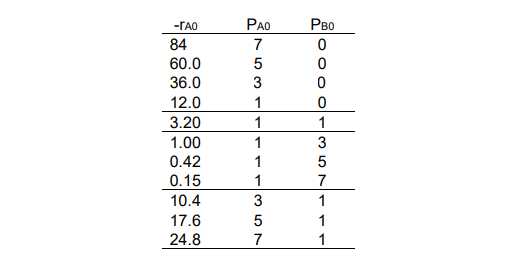
a) Write a full mechanism in the form of a system of reactions describing
adsorption, surface reaction, and desorption. Specify the type of adsorption
mechanism (molecular, dissociative) and reaction mechanism(s) (single site,
dual site, Eley-Rideal) determined to fit the data provided.
b) Determine the rate law(s) for each step (adsorption, surface reaction,
desorption).
c) Find the rate-limiting step and express the rate law in terms of unknown
constants and partial pressures.
d) Solve for unknown constants by fitting the model to the data computationally
(MATLAB is recommended) and then express the rate law solely in terms of
partial pressures. Demonstrate that the model fits the data.
Learning Outcomes
This coursework will fully or partially assess the following learning outcomes for this module.
Knowledge and Understanding
- A knowledge of reactor behaviour for homogeneous and heterogeneous systems. An understanding of reaction engineering so that students can apply the principles to the solution of relevant engineering problems and to the design of reaction processes.
In this coursework, you will use reaction engineering principles to model a heterogeneous catalysis process. This would be the first step towards designing a reactor for this process
Intellectual Skills
- Formulate dynamic material and energy balances to give a good representation of a chemical reactor for simple and complex reactions. Identify mixing and dynamic effects in chemical reactors. Construct models for non-ideal reactors. Make quantitative descriptions of heterogeneous reaction processes.
In this coursework, you will formulate a site balance for a catalyst surface in order to develop a quantitative model of a heterogeneous catalytic reaction process.
- Use a mathematical approach for design, optimisation, and control of chemical reactors.
In this coursework, you will use both symbolic and computational mathematics to develop this quantitative model.
Practical Skills
- Evaluate the performance of chemical reactors. Make provision for heterogeneous processes. Specify temperature profiles. Specify designs for homogeneous and heterogeneous reactors. Optimise reactor performance.
Are You Looking for Answer of This Assignment or Essay
The post In this part of the coursework, you will determine the mechanism and develop a rate law for a catalytic reaction from experimental data: Advanced Reaction Engineering, Coursework, LSBU, UK appeared first on Students Assignment Help UK.